New omicron variant BA. 2.75 erupts out of India
These images of the latest Covid variant have revealed a big risk in our immunity. And experts are warning, a new wave might be on the way.
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Science from Pfizer And Moderna = SPAMSunday Special: How India Foiled Pressure from Pfizer And Moderna, Confronted WHO on Excess Deaths Figure
Last year when the Covid pandemic was raging, Pfizer and Moderna offered their vaccines to India for $30 each on some drastic terms that the government refused to sign up for. Some months later, India told these vaccine makers that it was starting to export its own vaccines at $2.5 each, and they could buy them if they wanted to.
This year, India came together regardless of political affiliation to protest against the WHO’s estimate of 4.7 million excess Covid deaths in India. The Centre stuck to its stand that doing so was out of the global health body’s ambit and if it had to do so, it should have shown deaths per million, putting it on record at the World Health Assembly.
News18 spoke to top government sources who narrated these events in detail and how the Narendra Modi government decided to stand up to global vaccine giants in “national interest” and countered the WHO’s efforts “to put India down” despite India’s comparative better performance against Covid-19 than the West.
Pfizer and Moderna bargained, India went local
Rewind to last year around the time when the second wave was creating havoc in India as well as the rest of the world. Top government sources recall how there was immense pressure from all sides to buy Pfizer and Moderna vaccines, with the foreign manufacturers offering them at $30 each. “The government knew it would not be able to administer those expensive vaccines to each Indian,” a source said. But what bothered India more were their conditions.
“They wanted us to give a sovereign guarantee which meant that the vaccine that required -70 degrees Celsius storage, exported from the US, if any cold chain got disturbed (making the vaccine useless), the company had no responsibility. If anyone who got the vaccine faced any side effects or sued the company, these companies will not take any responsibility, rather it will be the responsibility of only and only the Indian government,” government sources said.
Also, as the vaccines were in emergency use and, meanwhile, if some major event took place (for instance, death or disability due to vaccine administration), these companies were seeking protection from India by asking that the Indian government fight cases in international courts. “Or if the company chooses to fight, India will bear their expenses. To ensure that such feuds would be taken care of by the Indian government solely, they also asked for a sovereign guarantee in which they sought the right to seize our properties,” the government sources further revealed.
It is then that India decided to call off these talks and union health minister Mansukh Mandaviya was sent on a four-day trip across the country to the factories of Biological E, Zydus Cadila, Bharat Biotech, and Dr Reddy’s. Mandaviya spent a day in each company and had a list of 25 questions for them to ascertain their strength, weakness, financial strength, and manufacturing capacity, sources say. The minister returned and the government arranged finances and raw materials, gave the companies advance payments, and helped them set up fill and finish capabilities. This led to an increase in the production of Indian vaccines by three times in just one month.
India then again invited the foreign vaccine makers asking them their possible vaccine delivery timelines, who said the first consignment could be given in November 2021. “We then told them that we will start our exports in October and that too for $2.5 per vaccine dose; if you need, you can buy. It was all about respect for our country which we couldn’t afford to let down. Their bargaining felt like selling our own country,” a source said.
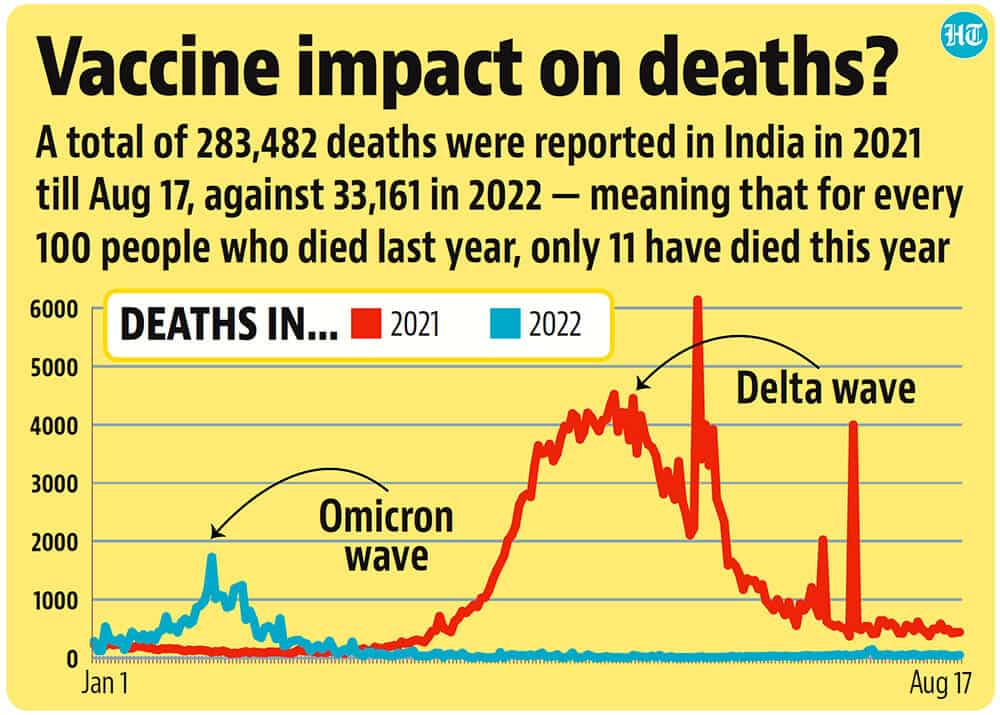
India has done 199 crore vaccinations through its ‘made in India’ vaccines and will be reaching the record 200-crore mark this month.
WHO saw India’s pushback
A situation again emerged this May when the World Health Organization (WHO) said that 4.7 million people had died in India due to Covid-19, far more than the Indian government’s official figure of about 5 lakh deaths. India was perplexed why the WHO had done meetings with countries for three months across the globe and if it was to divert attention from the ongoing events in the other countries where people were still dying in the third wave.
A source said the WHO’s mandate is to encourage and promote best practices, improve poor health facilities, provide access to health facilities, identify new epidemics, find out treatments, and make those treatments affordable and accessible. “But it seemed they had to prove, anyhow, that India had a higher number of deaths considering that globally, everyone was praising India for good work in handling Covid-19. Just to put us down, the WHO did this exhaustive exercise,” a top government source told News18.
Sources said the government had given the WHO all the facts — that India had a proper system of registering births and deaths and the same has been compulsory since 1969, resulting in 99.9% of deaths being registered. The register said that in the Covid year, an excess of 9 lakh deaths had been reported against the regular average. Out of 9 lakh, 6 lakh deaths were attributed to Covid while the rest were due to non-registry, India told the WHO.
The Centre was in the midst of a three-day meeting with the state health ministers when the news of the WHO’s estimate of 4.7 million deaths in India came out. Union health minister Mansukh Mandaviya told all the state ministers to forget their political affiliation and parties because “it is about the self-respect of India”, a source said. A unanimous resolution was signed by the health ministers of all states with Mandaviya assuring them that he would go to the World Health Assembly and tell them that whatever had been done was incorrect and clearly out of the WHO’s mandate.
“Even if the WHO had to do so, it should have shown deaths per million, but it did not,” a top government source said.
India felt that given its 1.3 billion population, huge diversity, and still being able to control Covid-19, this was something many people were not ready to accept. “This is while several other countries continue to jostle with Covid-19,” the source added.
Sunday Special: How India Foiled Pressure from Pfizer, Moderna, Confronted WHO on Excess Deaths Figure
India felt that given its 1.3 billion population, huge diversity, and still being able to control Covid-19, this was something many people were not ready to acceptwww.news18.com
WHO = Weed Hukkah and Opium organization
If you are taking this covid vaccine, beware of potential allergic reactions: EU
The typical reactions to Novavax COVID-19 vaccine are mild, including arm pain and fatigue. But there are possibilities of a rare risk, heart inflammation

Amid monkeypox, Covid spread, concerns over ‘tomato fever’: Know the symptoms
So far, tomato fever has been reported in Kerala, Karnataka, Tamil Nadu and Odisha. Although authorities are on alert over its spread, they have asked people not to create panic.
India pushes makers for bivalent Covid vaccines
Indian government scientists are in discussions with domestic vaccine makers for a bivalent Covid-19 dose, similar to the approach taken by US pharma company Moderna for its shot that became the first updated coronavirus vaccine in the world this week, people aware of the matter said, adding that approvals for such products will be fast-tracked.The department of biotechnology in touch with vaccine makers for what officials call a second-generation vaccine, and at least two companies — Serum Institute of India (SII) and Bharat Biotech — confirmed they were working on prototypes.

“We are looking at a bivalent vaccine that can provide an immune response against two antigens through a single vaccine,” said a senior official from the department, requesting anonymity.
The official added that the department was in talks with five pharma companies, as well as research organisations. “It is too early to give out much details, but considering that the newer variants of Covid-19 now have a greater immune escape, there is a need for updated vaccine,” the official added, refusing to divulge the name of the companies that were in talks with the department.
A bivalent vaccine is designed on the basis of two pathogens. Moderna’s mRNA 1273.214 for instance is a 50-50 combination of its 2020 product and a new one based on the highly resistant Omicron variant, allowing it to create immunity that protects against both versions of the virus.
The Omicron variant’s lineages are now dominant across the world, becoming a de-facto new foundation for the Sars-CoV-2’s ongoing evolution. The World Health Organization and an international coalition of health regulatory agencies first sought new vaccines to counter such variants in January.
People aware of the matter said an update to an existing vaccine will be fast-tracked. An official from the office of the Drugs Controller General of India (DCGI) said approvals for updated vaccines will in themselves not be a time-consuming process because basic formulations are already in place and have gone through the compliance process once.
“The priority of the government currently is on Covid-19 vaccines and their newer versions. So, it’s plain logic that approvals will be fast-tracked accordingly,” said the official.
Officials from SII and Bharat Biotech, the producers of the two doses most widely used in India’s Covid-19 vaccination till now, said they were working on such doses.
An SII spokesperson said the Pune-based company is working with the American vaccine developer Novavax to update the dose being made and sold in India under the brand name Covovax. “The existing formulation will be tweaked to incorporate the new variant and because this is not going to be a new vaccine per se, we will not take much time to get approvals,” the official said, without divulging details about the timeline.
An official from Bharat Biotech said the company was developing a “variant proof” vaccine and immunogenicity data was being studied.
At present, Covishield (produced by SII), Covaxin (developed by Bharat Biotech), Biological E’s Corbevax, Gamaleya Institute’s Sputnik, and SII-made Covovax are in use in India. The regulator has also approved doses by Moderna and Pfizer-BioNTech but these have not been available in the country.
Earlier this week, UK authorities cleared a Moderna dose that the company adapted to create immunity against the Omicron variant. The vaccine is a 50-50 formulation of Moderna’s 2020 product, designed on the Sars-CoV-2 variant that first spread around the world, and of a new dose developed using the blueprint of the Omicron variant. Other companies, such as Pfizer, too are working on similar updates.
The need for Covid-19 vaccines to be updated was first visible in January when the Omicron variant quickly spread across the world, displacing all other variants with its increased resistance to antibodies elicited from vaccines and past infection — which otherwise had blunted the growth of versions like Alpha and Delta.
Recently, scientists and global health bodies have flagged that the protection from severe diseases conferred by booster doses is “transient”, making updated vaccines necessary instead of a repeated, frequent boosters.
“The incremental benefit of a second booster dose is likely lower compared to primary series and first booster doses. The number needed to vaccinate to avert one death with a second booster is higher compared to that for primary series,” WHO said in a statement on Thursday regarding the use of second booster doses.
“The impact of currently available vaccines on reducing symptomatic illness and transmission in the context of Omicron is modest. The effects of a second booster dose on restoring VE against symptomatic illness will be transient and likely not last longer than a few months,” it added.
Several countries are now offering second booster doses. India is at present offering the first booster doses to people who have completed six months since their full primary vaccination course.
Gagandeep Kang, virologist and professor at Christian Medical College, Vellore, said it would be good to have a broader ‘Sars-CoV-2’ vaccine that provides protection against a range of Sars-CoV-2 variants. “There could be a single antigen that has a broad response against a range of Sars-CoV-2 or have a universal coronavirus vaccine that provides immunity against current and future mutations. It would definitely be a good idea to develop the science for both,” Kang said.
“The goal is to have an immune response against Covid which is both broad and long-lasting because we don’t know what the next variant is going to be. The aim should be to add as many different antigens as possible, add antigens beyond the spike protein. Ultimately, scientists would want to develop a ‘pan coronavirus vaccine’, which would be broadly protective against all the variants but also other types of Sars-CoV viruses,” said Soumya Swaminathan, chief scientist at the World Health Organisation (WHO), in a tweet on Wednesday, citing an interview she gave to a news channel.

India pushes makers for bivalent Covid vaccines
At least two companies — Serum Institute of India (SII) and Bharat Biotech — confirmed they were working on prototypes.

Here’s all that India did right to manage Covid-19 crisis, according to World Bank
The paper highlights the innovations that India came up with to tide over the crisis, including efforts to develop the domestic market for essential Covid-19 commodities and lifesaving equipment
India's first intranasal Covid vaccine by Bharat Biotech gets DCGI approval
Health min calls it to be a ‘Big Boost to India's Fight Against COVID-19’
COVID raises risk of long-term brain injury, large U.S. study finds
People who had COVID-19 are at higher risk for a host of brain injuries a year later compared with people who were never infected by the coronavirus, a finding that could affect millions of Americans,...
Is this really plausible? I mean, if you were a pregnant woman would you even volunteer for a drug trial?

